Abstract
Summary: Rabies encephalitis is perhaps one of the few infectious diseases that command attention and fear not only from the layman but also from physicians. The unique mode of transmission, the virtually exclusive neurotransmission shown by the agent, and the complete hopelessness of the established disease sets rabies apart from other zoonoses transmitted to man. Rabies encephalitis is a fatal disease and its diagnosis is usually based on the clinical presentations and findings. Hence, imaging in rabies is seldom done, and imaging findings in rabies encephalitis have rarely been described. We present the imaging findings in two confirmed cases of rabies encephalitis in which antemortem diagnosis was obtained by corneal impression smears showing the presence of viral antigens. The differential diagnosis of the imaging findings as well as the role and the relevance of imaging in the diagnosis of this disease are discussed. The current literature on the subject is also reviewed.
Rabies encephalitis is one of the oldest communicable diseases known to man (1). It is an acute infection involving the CNS in humans and other mammals caused by an RNA virus of the rhabdovirus family. Transmission of the disease is through the bite of dogs and wild animals. Other forms of transmission are through inhalation in bat-infested caves and in laboratory settings (2, 3). In rare instances, human-to-human transmission by infected corneal transplants has been reported (4). Human rabies presents in two forms: encephalitic and paralytic. These forms are analogous to the furious and dumb rabies seen in dogs. It has long been thought to be an invariably fatal disease with few reports of survival (5). The fulminant course of the disease makes imaging in these patients uncommon. We describe the imaging features in two patients with rabies encephalitis. Both patients were infected through dog bites, and corneal impression smears obtained from both patients showed the presence of the viral antigens on fluorescent microscopy. The pathophysiology of the disease and the radiologic-pathologic correlation of the findings are discussed.
Case Reports
Case 1
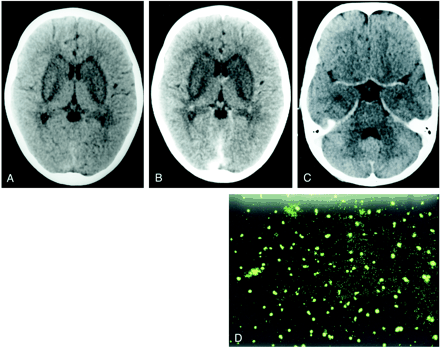
A 6-year-old boy presented with sudden onset of generalized seizures and altered consciousness 2 weeks after being bitten by a dog. On physical examination he showed increased tone in all four limbs, with hyperreflexia and extensor plantar responses. Plain and contrast-enhanced CT studies of brain showed nonenhancing, symmetrical hypodensities involving both basal ganglia (Fig 1A and B). No areas of altered attenuation were seen elsewhere in the brain (Fig 1C). In view of the clinical history, rabies encephalitis was considered a possible diagnosis, and was confirmed by detection of the viral antigens in corneal impression smears on immunohistochemistry. The viral antigens targeted by the fluorescent-labeled antibody appeared as apple-green fluorescent bodies under the fluorescent microscope using the fluoroscein isothiocyanate filter (Fig 1D). The patient was managed conservatively and died within 4 days of admission. Postmortem examination was not performed.
Case 1: 6-year-old boy with generalized seizures and altered consciousness.
A–C, Plain (A) and contrast-enhanced (B and C) CT scans show nonenhancing bilateral basal ganglia hypodenstities. No other areas of altered CT attenuation were seen. There was no evidence of enhancement on administration of contrast material. No areas of altered attenuation are seen in the brain stem (C).
D, Photomicrograph with immunoflourescent staining of the corneal impression smear shows bright apple-green viral nucleocapsid antigen targeted by the virus-specific fluorescent-labeled antibodies and seen using fluorescein isothiocyanate filter.
Case 2
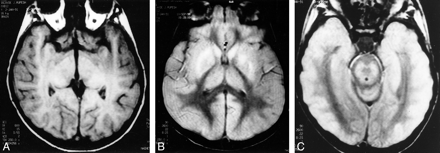
A 30-year-old man presented with acute onset of altered consciousness and drowsiness 3 weeks after being bitten by a dog. He had had prodromal symptoms of fever, headache, and lassitude for 4 days. On physical examination, the patient was unconscious, with flaccid paralysis in all four limbs. All reflexes were absent bilaterally. TI- and T2-weighted MR images of the brain revealed hyperintensity in the globus pallidus, putamen, and thalamus bilaterally (Fig 2A). Proton density–weighted axial images showed hyperintensity in the head of the caudate nucleus, globus pallidus, putamen, and pulvinar of the thalami bilaterally (right more than the left) (Fig 2B). No abnormal signal was detected from the brain stem (Fig 2C). Owing to the patient's history, paralytic rabies encephalitis was diagnosed and subsequently established antemortem on the basis of findings on the corneal impression studies similar to the first case. The patient gradually deteriorated and died within 3 days of admission. Postmortem examination was not performed.
Case 2: 30-year-old man with altered consciousness and drowsiness.
A, Axial spin-echo T1-weighted MR image (600/15 [TR/TE]) shows hyperintensity in the globus pallidus and putamen bilaterally. Minimal hyperintensities are also seen in the thalamus bilaterally.
B, Axial spin-echo proton density–weighted MR image (2600/22) shows hyperintensities in the heads of the caudate nuclei bilaterally. Hyperintensities are also seen in the globus pallidus and putamen bilaterally (right more than the left).
C, MR image at the level of the brain stem shows no abnormal signal.
Discussion
The term rabies is derived from the old Indian root word rabh, meaning to make violent (1). The disease is also known as hydrophobia or aquifuga (1). Rabies encephalitis is an acute CNS infection caused by a type of RNA virus of the rhabdovirus family. Transmission to humans is mainly through bites of infected rabid dogs, cats, bats, and other wild animals. Other modes of transmission are through inhalation, by contact of infected saliva with an open wound or mucous membrane, and via infected corneal transplants (2–4).
The incubation period of rabies is typically 2 to 8 weeks (6, 7), although it varies with the type of infecting strain, and is found to be inversely related both to the size of the inoculum and to the proximity of the site of the bite to the CNS. The viruses are usually introduced deep into the soft tissues by an animal bite and infect the muscle, possibly through an affinity for nicotinic cholinergic receptors. They replicate in the muscle before reaching the CNS, either through the neuromuscular spindles or the motor end plates (6). Alternatively, the virus may directly affect the sensory nerve endings of the superficial soft tissue. The passage of the virus to the CNS occurs axonally through retrograde axoplasmic flow of approximately 12 to 24 mm per day until the virus reaches the next neuronal cell body (8).
In the CNS there is an initial proclivity toward infection of the gray matter. Tirawatnpong et al (7) suggested involvement of the neuroglial cells along with the neurons. Once inside the CNS, rapid dissemination of the virus takes place and rapid progressive encephalitis ensues (8). In paralytic rabies, the medulla and the spinal cord are mainly involved by extensive neuronal damage and inflammation, whereas in the encephalitic form, it is the brain stem and the cerebrum, particularly the limbic system (9, 10). Involvement of the basal ganglia and the thalamus is usually seen late in the disease (6, 7). The early localization of the virus in the limbic system, with cortical sparing, correlates clinically with behavioral and emotional changes seen in an alert and cognitively intact patient (6). Rabies virus reaches the CNS early in the disease and returns to the periphery late in the process by intraaxonal transport (6). This centrifugal spread of the rabies virions is observed late in the disease. There is an unexplained tropism for the salivary glands and lacrimal glands, with infectious rabies particles appearing in the saliva, tears, and tracheobronchial secretions (6).
Clinical illness begins upon arrival of the virus into the CNS. Human rabies may present in one of two forms: encephalitic and paralytic (7). In encephalitic rabies, the initial symptoms are nonspecific, such as fever, malaise, anorexia, cough, and pain or paraesthesia at the bite site. Subsequently, patients develop hydrophobia, aerophagia, hypersalivation, hyperirritability, hyperactivity, and priapism. Neurologic symptoms, such as seizures, agitation, and alternating mood swings, often occur. Paralytic rabies encephalitis causes more diagnostic problems, as the clinical symptoms of hydrophobia and aerophobia are present in only half these patients (7). Moreover, the initial presentation with flaccid paralysis resembles Guillian-Barré syndrome. The pathogenesis of paralytic rabies is poorly understood. Host immune response may be one of the factors deciding the clinical outcome (7). Paralytic rabies appears to correlate with absence of significant immune response to the infection and diminished number of peripheral blood B lymphocytes. Both forms of the disease are invariably fatal and most patients die within 10 days of the onset of neurologic symptoms. Patients usually succumb to failure of basic central vegetative functions, although death may also be due to concomittant rabies myocarditis (6).
Because the disease has a rapidly fulminant course, imaging of these patients is uncommon and difficult. Hence, the imaging findings in naturally acquired rabies have received little attention in the literature. A CT scan may show focal or diffuse areas of decreased attenuation in the basal ganglia, periventricular white matter, hippocampus, and brain stem. Pontine hemorrhages have also been reported. Diffuse cerebral edema may be seen in advanced cases (11). Murthy (12) reported findings of multiple areas of white matter on MR images, including in the brain stem, cerebellar peduncles, and both cerebral hemispheres, in a case of vaccine-induced acute demyelinating encephalomyelitis. Sing and Soo (11) reported finding a narrowing in the supraclinoid internal carotid and terminal basilar arteries on a cerebral angiogram. This was postulated to be due to transient arterial spasm induced by the viral infection.
Both our patients had identical involvement of the basal ganglia bilaterally, seen as areas of hypodensity on CT scans and as areas of hyperintensity on T1- and T2-weighted MR images. In one of the patients, MR images also showed hyperintensities in the pulvinar of the thalamus bilaterally. The MR features suggested extracellular methemoglobin, perhaps due to the late presentation of the patients, when the disease was well established; however, we do not have pathologic proof of this. We do not believe that the hyperintensities were related to hydrated calcium because of the short duration of the disease in both patients. Similar imaging findings are seen in many conditions, such as infectious viral encephalitis, cholinergic poisoning, toxic encephalopathy (carbon monoxide, methanol, hydrogen sulphide, cyanide), severe hypoglycemia, extrapontine myelinosis, hypoxic/ischemic insults, and hemolytic-uremic syndrome; and in unusual conditions, such as Wilson disease and Leigh disease. Such a finding in patients with a history of an animal bite and clinical sequelae, as in our patients, is highly suggestive of rabies encephalitis. Both our patients had incurred a dog bite, and corneal impression smears obtained from both of them showed the presence of the viral antigens on fluorescent microscopy. The atypical sparing of the brain stem cannot be explained in either of the patients.
Gross pathologic specimens of the brain in rabies show scattered haemorrhages, necrosis, and surrounding edema in the cerebral gray matter, predominantly involving the basal ganglia and the brain stem. Histopathologically, the changes seen in the neuron are of necrosis. There can be associated degeneration of the axis cylinder and demyelination. Whenever the neuronal changes are severe there may be marked hyperemia with petechial hemorrhages, especially in the thalamus and the subependymal neuroglial tissue (8). This pattern of pathologic involvement correlates well with the imaging findings seen in both our patients, with nonhemorrhagic infarction seen as an area of hypodensity involving the basal ganglia in the first patient and hemorrhagic infarction of the basal ganglia and the thalamus in the second patient.
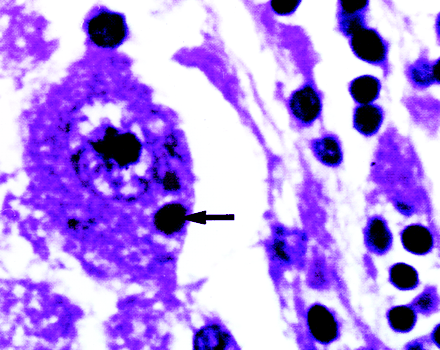
Neuropathologic specimens in rabies show intraneuronal Negri inclusion bodies. Classic Negri bodies are round or oval, discrete, sharply demarcated eosinophilic intracytoplasmic bodies. They are most commonly seen in the pyramidal cells of the hippocampus, cerebral cortex, and Purkinje cells (13, 14). They may be anywhere in the cytoplasm or in its dendrite, and there may be more than one in a single cell (Fig 3). Negri thought them to be a stage in the life cycle of the virus; however, immunofluorescent staining has resulted in detection of the viral antigen within the Negri body (13).
Histopathologic section of the brain from a different case shows the presence of intracytoplasmic Negri bodies (arrow) in the cortical neuron (hemotoxylin-eosin, original magnification ×400)
Premortem diagnosis is usually made by performing corneal impression smears, nuchal skin biopsies, or biopsies of the oral or nasal mucosa. These techniques are dependent on the centrifugal transport of the rabies virus to the peripheral nerves late in the disease and hence show negative findings in the early stages of the disease. A corneal impression smear is made by vigorously pressing a microscope slide against the eyeball. The impressed cells are then fixed, stained, and read using immunofluorescent techniques, which are performed using a polyclonal antinucleocapsid antibody or an IgG against rabies intracytoplasmic nucleocapsid antigen (6, 15, 16). Specificity of this technique is 100%, but the sensitivity varies for different specimens (7). When rabies encephalitis is suspected, special management of the tissues obtained for the histopathologic study is necessary. Use of face masks, gowns, and gloves by all the personnel in contact with the patient and the tissue sample is necessary. Saliva, CSF, and tissues should be handled and disposed of with precautions. In addition, all known or suspected human contacts (premortem or postmortem) should receive postexposure prophylaxis (6).
Conclusion
The diagnosis of rabies is essentially clinical. Patients may present with a wide variety of symptoms, ranging from widespread CNS excitation to flaccid paralysis. Although a differential diagnosis based on imaging findings of basal ganglia involvement is remote, in the appropriate clinical context, encephalitis due to rabies needs to be considered. In diagnosed cases, managing of patients and handling of tissues require special precautions.
Footnotes
↵1 Address reprint requests to Dr. Hemant Parmar, Department of Radiology, King Edward VII Memorial Hospital, Parel, Mumbai 400 012, India.
References
- Received May 23, 2000.
- Copyright © American Society of Neuroradiology